Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
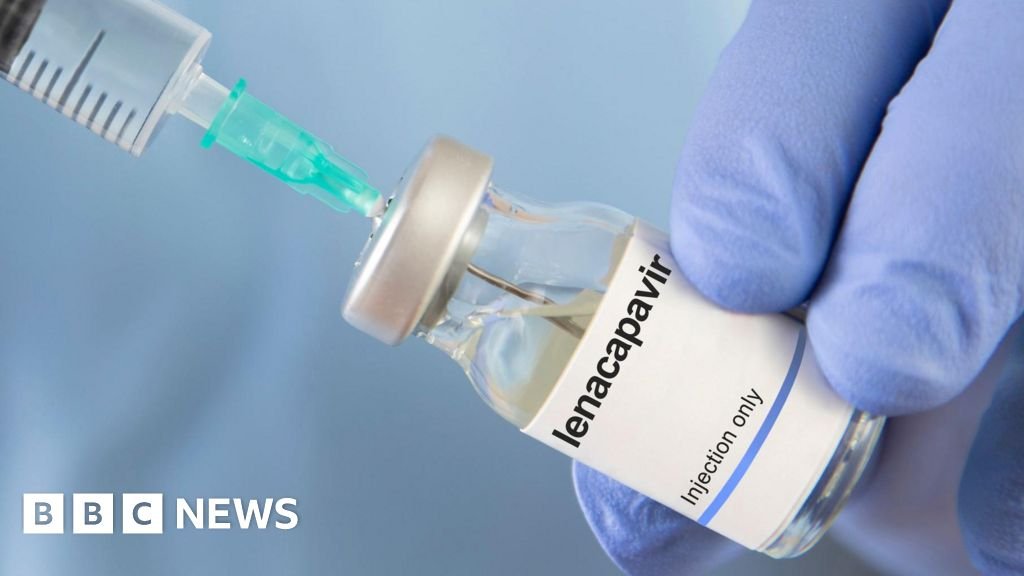
A new HIV prevention medicine will be provided at a lower price in over 100 low-income countries within two years, which is expected to give millions of access to breakthrough treatment and potentially bring the world closer to the termination of HIV/AIDS epidemic.
The drug, called Lenacapavir and is administered by injection, should be implemented at the end of this year, priced at $ 28,000 (20,000 pounds) per person annually.
But the message on Wednesday promises to reduce this price to only $ 40 – about 0.1% of the initial costs. The lower price version will be introduced in 2027 in 120 low and medium-sized countries.
Scientists say the drug stops the virus from replicating inside the cells.
The main deal to provide more expensive antiretroviral drugs for people with HIV in developing countries was mediated by former US president Bill Clinton after negotiations with pharmaceutical companies.
The agreement, announced on Wednesday, was reached between the Clinton Foundation in partnership with the Gates Foundation and other groups, including the South Africa Research Institute, Whits Ri.
Lenacapavir gave impressive results from the tests and in July received formal support from the World Health Organization for HIV Prevention.
The injection is taken twice a year and provides six months of protection against HIV infection at a time.
Experts say injection injectors such as Lenakapavir could help reduce new infections in populations that are most vulnerable, including girls from adolescents and young women, LGBT people, sexual workers and those who use drugs.
We hope that he will replace the current form of preventive HIV medicine, know as preparations or prevention before exposure, which is accepted orally and also costs $ 40 per person every year.
The pills are taken daily, which can be difficult to consume patients and can carry stigma in certain societies. The daily dose also makes it more difficult to access. According to the Gates Foundation, only 18% of those who could benefit from PREP are currently available.
Lenacapavir has already been approved by the US Food and Drug Administration and the European Commission this year.
Last year, June, the American drug company Gilead announced that the HIV test Lenacapavir had a 100% success rate.
The new, generic version is still in anticipation of regulatory approval, but we hope it will be available within 18 months.
One study says increasing access to injection to only 4% of the population can prevent up to 20% of new HIV infections.
The drug can be used to protect people from catching the virus, but also to treat those who have it, according to scientists.
The announcement comes against the backdrop of a tumultuous year of global health, especially the fight against HIV and AIDS, after a cut of cuts from the foreign assistance program of the US government, USAID, under the Trump administration.
According to UNAIDS, more than 40 million people currently live with HIV.
Despite the huge steps made to reduce the global HIV rates and the deaths associated with AIDS since 2000, about 1.3 million people were still infected with HIV last year, and over 600,000 people were killed by AIDS.
South Africa remains the country with the largest number of HIV cases, with nearly eight million people living with the virus. This will be one of the countries that have access to the new, cheaper medicine.
Additional reporting from Akisa Wandera in Nairobi