Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124

In science, there is an amazingly long list that we have not yet discovered yet but they still use it. It was unexpectedly in the case of lithium-ion batteries — a power source of electric vehicles and various portable electronics-where scientists knew what the process was but exactly how it worked.
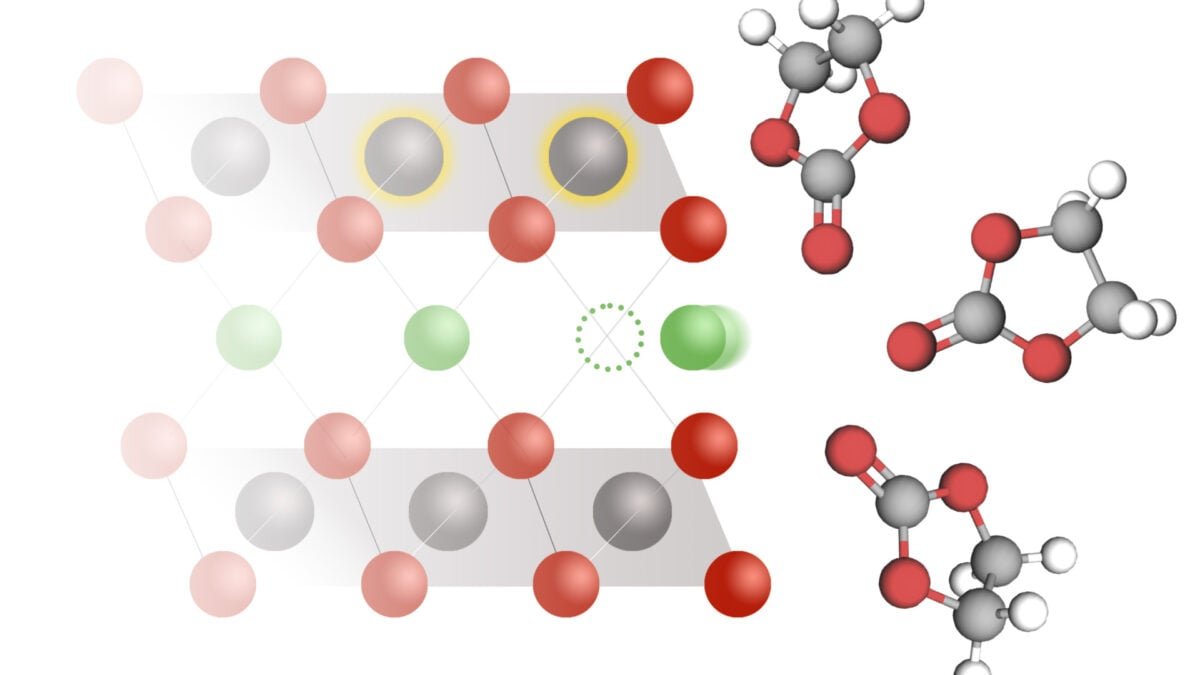
Fortunately, MIT scientists have found the answer. Ay Science Paper published on October 2, researchers describe a model that depicts how combined ion-electron transfer (CIET), an electrode where an electron travels with an ion, can explain a lithium ion, lithium ion battery. According to researchers, insight “can guide the design of the lithium-ion batteries more powerful and quick charging.”
A simple lithium-ion works through a chemical system called battery intercation. Basically, during the battery discharge, the lithium ions dissolved in the electrolyte solution sell themselves inside a solid electrode. When the ions return to “D-Intercate” and electrolyte, the battery charge.
The interconnection rate operates everything from the battery net strength to its charging speed – because researchers thought it was essential for better understanding processes, explaining the paper.
Previously, scientists believed that the lithium inter -cell was powered by a model on the battery electrode describing how quick lithium ions could spread between electrolytes and electrodes. However, the actual tests were not fully matched with the prediction of the model, the researchers suggested that there could be another option here.
For new studies, researchers prepared a combination of more than 50 electrolytes and electrodes to straighten things once and for everyone. Like the previous tests, they found large -scale inconsistencies between actual data and models. So instead, the team came up with several options that could explain what they were seeing.
Finally, they decided on a model based on this concept that lithium ions can only enter an electrode if it travels from an electrolyte solution to the electron-transfer the ion-electron transfer. This electronic chemical pair makes it easier to interconnect, researchers explain and maths on the back of the CIET fit well.
“The electronic chemical action is not the lithium insert, which you can think is the main thing, but to reduce the hard elements of the lithium hosting it is actually transfer,” Martin Bajant, the study co-authors and a mathematician of MIT, said, MIT newsThe “Lithium is interclated as the electron is transferred and they facilitates each other.”
Not only that, the researchers also accidentally discovered that the combination of electrolytes influences the inter -rate switching up. They have explained that follow-up investigations can unravel more efficient ways to create strong, quick battery.
Bajant said, “The reactions we have been able to do by this work are to be quick and more controlled, which can speed up charging and discharge,” said the Bazan.