Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
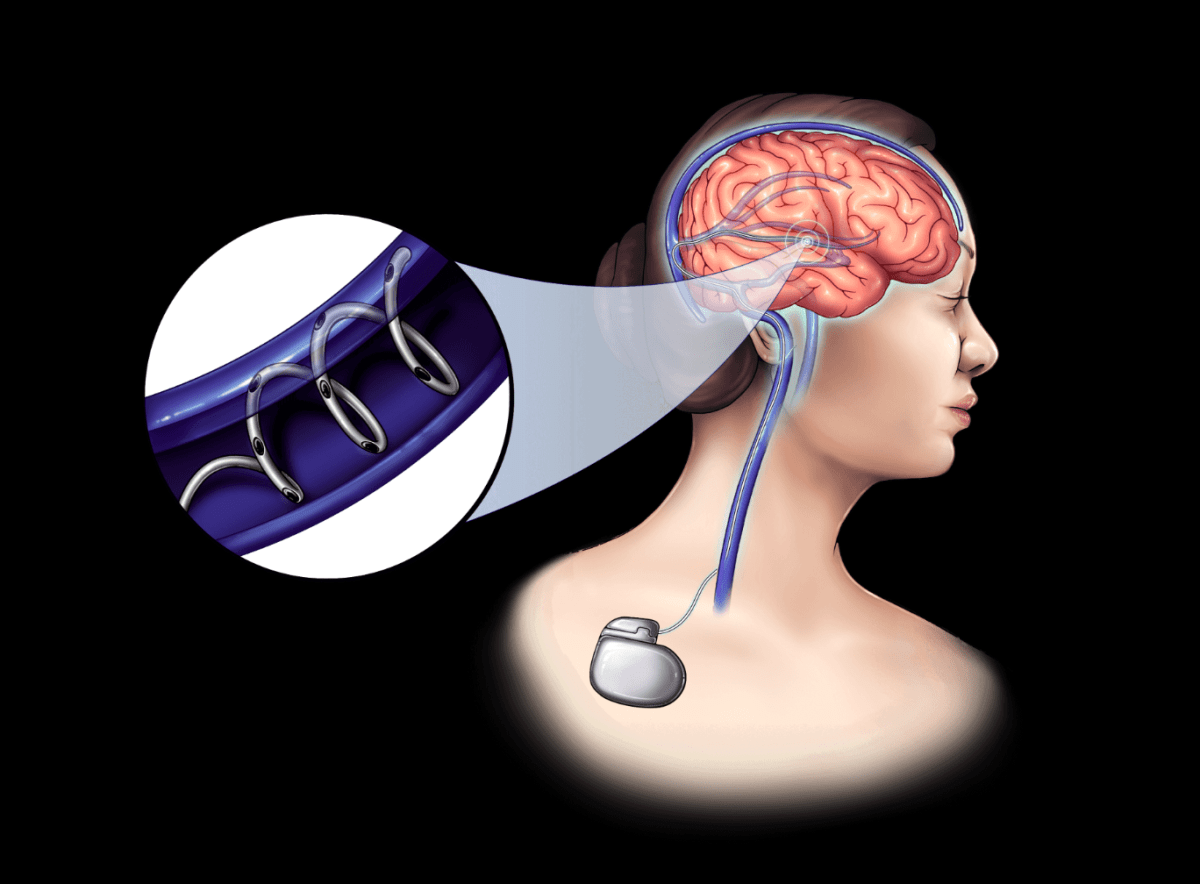
You can’t start a company without a healthy dose of courage, and this is certainly the case Neurobionics. The MIT-spinout thinks it could one day improve the lives of millions of people living with neurological conditions like depression, epilepsy and Parkinson’s disease.
Celebrity investor Steve Jurvetson of Future Ventures said that if all goes well for the 18-month-old outfit, its system could address “the peripheral nervous system for pain, incontinence and a bunch of other applications.”
How? with what In contrast to this exotic ambition, the technology of neurobionics is small. Specifically, Neurobionics aims to use what it has created — bioelectric fibers the width of a human hair — to deliver neuromodulation therapy through the brain’s blood vessels, a method similar to stent placement.
The fibers are powered by a fairly standard implantable battery shaped like an AirPod case, designed to last five to 10 years, and used by other medical device makers for spinal cord stimulation, among other things.
It’s a pretty nifty alternative to drilling a hole in someone’s skull, as has long been the procedure for deep brain stimulation. Traditionally, when certain disorders do not respond to medication, metal electrodes are implanted in the brain to generate electrical impulses and control abnormal movements.
Neurobionics devices are not only less invasive; The company is using carbon nanotubes instead of thin-film platinum or iridium oxide, which are common materials for these electrodes. Although metals are minimally toxic and conduct electricity well, they can dissolve, limiting their lifespan and causing tissue damage. Carbon nanotubes, on the other hand, are cheap, obviously last longer, and they make getting an MRI much easier. (Among other things, metal can create bright spots on MRI images, making the brain difficult to see.)
According to MJ Antonini, CEO of the Cambridge, Massachusetts-based startup, the whole shebang is the result of 10 years of research on fiber technology at MIT. He co-founded the company while still a student at the school, where he acquired three patents that gave MIT a small stake in the business.
He took an interesting route from point A to point B. On the Zoom call, as Antonini shows a coiled version of the barely visible fiber, he explains that he has earned doctorates from both Harvard and MIT through a 55-year-old. Known as the Harvard-MIT Program in Health Sciences and Technology.
Calling it a special program they don’t advertise for the wrong reasons, Antonini said his studies included two years of medical school at Harvard, followed by medical engineering and medical physics at MIT. After that, he decided to “go beyond the core [research] paper” and “build a real product and a real medical company.”
In fact, Antonini, who is French, said he stayed on as a postdoctoral researcher for a few more years to think about how to bring that portfolio of technologies into the real world. He eventually left school in early 2023 with Nicky Driscoll, an associate postdoctoral researcher at MIT and today the CTO of Neurobionics.
It will take a long time to know what their fiber technology will be. Like Zurvetson, Antonini asserts that eventually, neurobionics’ bioelectronic fibers could be used in a full spectrum of applications, including drug delivery, ablating tissue in the brain, and treating conditions related to the spinal cord and peripheral nervous system.
“Finally” is still some time away, though. For now, the organization has closed $5 million in funding led by Dolby Family Ventures, with participation from Future Ventures, GrayMatter Capital and other backers, and will use the capital to complete work on its clinical device.
Once complete, the next step will be to try to demonstrate the safety and efficacy of pigs, which are very similar to humans in terms of anatomy, physiology and genetics. The FDA will then review that work, after which Neurobionics can apply for an investigational device exemption (IDE). It can then begin its first-in-man initial feasibility study.
When asked if its technology could actually come to market, Antonini hesitated for a moment before suggesting 2030.
Of course, he wouldn’t work at a startup if he didn’t think it could navigate these next steps.
Patient investors like Jurvetson will help. “Deep brain stimulation has been shown to work in stroke, epilepsy, Parkinson’s, Alzheimer’s, chronic pain, tremors and more.” “But 99% of people who could benefit are rejected precisely because it requires large open brain surgery with needles inserted deep into the brain,” Jurvetson wrote in an email.
As far as Jurvetson is concerned, technology like NeuroBionics’ has widened that market — in part because today there is a concentrated pocket of large and advanced hospitals that offer the surgery.
The “application domain” for the startup’s “minimally invasive stent,” encourages Jurvetson, is “huge.”