Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
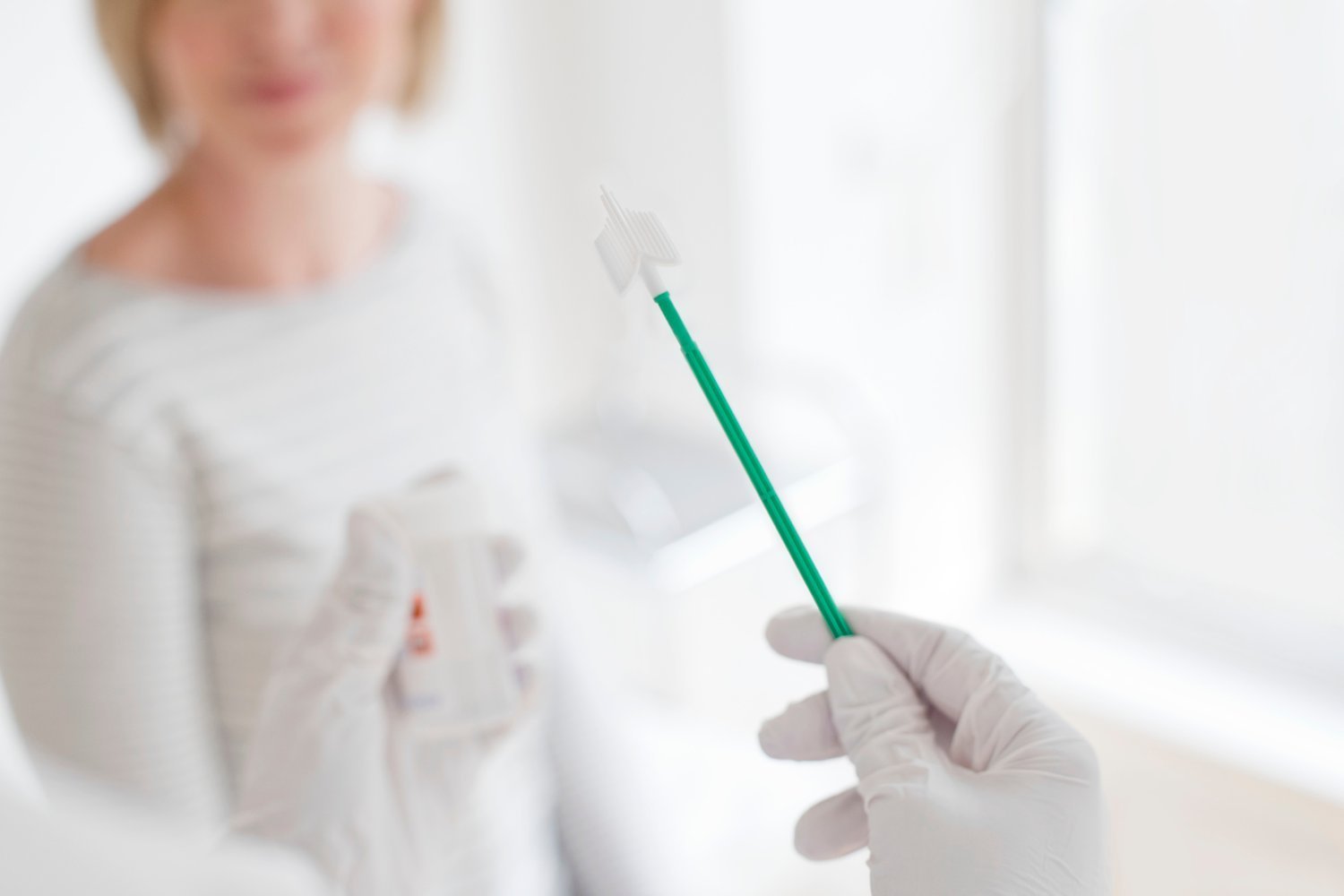
PAP smirms are an important tool to prevent uterine cancer because they can detect cell abnormalities before cancer. Unfortunately, a lack of comfort with the procedural barrier or the process of the process means that many people are not accepting it. However on Friday, FDA approves the first non-home test for uterine cancer screening This is a less aggressive and potential more accessible option for pap smayers.
The new FDA-approved test was developed by San Francisco-based startup til health. Like the Traditional Pap Smiers, Till Health Examination for Human Pypelomavirus or HPV, which is virtually caused by all uterine cancer. However, it requires a vaginal swab instead of inserting a spongle to scrap the scrap cells from the uterus.
In a statement, who is the CEO of Til Health and the co-founder who is Igan, D Authorization is “not just about an innovative product – it’s finally giving women an option that makes sense for their lives – something that can be done fast and comfortable at home”
Igan also said, “Because we are easy to take care of women we are healthy and we help them for those who depend on them every day.”
In the United States, almost 11,500 new suit Cancer cancer is diagnosed every year and 4,000 people will die. However the cancer of the uterus may “eliminate”[d] In our lifetime, “Dr. Alexe Right told the New York Times the director of the gynecology oncology of the Dana-Farmer Cancer Institute.” We have tools to do this. We do not only have HPV vaccine or uterine cancer screening and often cannot reach high risk people. “
According to the American Cancer Society, Cervical cancer More than half of the United States has dropped from the mid -1970s, though it has recently closed. A 2022 Study It has been found that 23% of people in 2019 were behind their screening compared to 14% in 2005. Although the reasons are different, a Study It has been found that the lack of insurance and expenditure was the common cause. Also, the rate of cervical cancer More than 25% In the countryside and the death rate is 42% higher, perhaps due to lack of resources and healthcare suppliers. Discomfort is also a major obstacle to some. In 2023, many participants in Michigan Medicine Studies D The room that sits on the kit “provides a comfortable, convenient and flexible choice without trauma.”
Home-A-Home screening is an effective alternative to the screening interval. One Study Mailing kites to people for screening of uterine cancer have increased by 50% compared to only normal care. Also, they are Exactly as correct As clinical examination. (Till Health also has a Self-service Study.)
Til health plans to make the kites available next month, starting in California. The company is also coordinating with the chief insurance suppliers in the hope of covering the cost. In addition to requesting examination via the til health platform, people can adjust follow-up care if needed.