Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
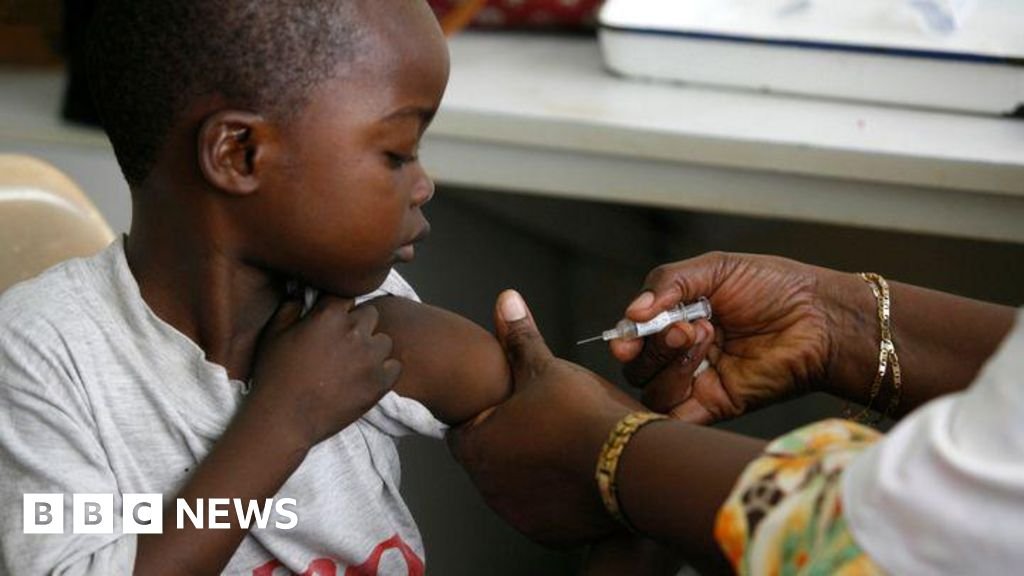
The first malaria treatment, suitable for babies and very young children, is approved for use.
It is expected to be implemented in African countries within weeks.
So far, there have been no approved malaria medicines, especially for babies.
Instead, they have been treated with versions formulated as bigger children, which is the risk of overdose.
In 2023, the year for which the most data are available – malaria was associated with about 597,000 deaths.
Almost all the deaths were in Africa, and about three quarters of them were children under five.
Malar treatments for children exist, but so far there has been no specific for the youngest babies and young children, who weigh less than 4.5 kg or about 10 pounds.
Instead, they are treated with medicines intended for older children.
But this is risks as the doses for these bigger children may not be safe for babies whose liver functions are still developing and whose bodies treat drugs differently.
Experts say this led to what is described as a “difference in treatment”.
Now a new medicine, developed by Novartis, has been approved by the Swiss authorities and is likely to be implemented in regions and countries with the highest rates of malaria within weeks.
Novartis plans to introduce it largely not with profit.
The Chief Executive Officer of the company SAC Narasimhan says this is an important point.
“For more than three decades, we have remained the course in the fight against malaria, working mercilessly to provide scientific breakthroughs where they are most necessary.
“Together with our partners, we are proud that we have gone further to develop the first clinically proven malarial treatment for newborns and young babies, guaranteeing that even the smallest and most vulnerable can finally get the care they deserve.”
The drug, known as Baby or Riamet Baby in some countries, has been developed by Novartis in collaboration with Malaria Venture (MMV) medicines, based in Switzerland, a non -profit organization originally supported by the British, Swiss and Dutch governments, as well as the River and Foundation.
Eight African countries have also participated in the assessment and tests of the drug and are expected to be among the first to have access to it.
Martin Fichet, CEO of MMV, says this is another important step on the way to ending the huge fee taken from malaria.
“Malaria is one of the most deadly diseases in the world, especially among children. But with the right resources and focus, it can be eliminated.
“Coartem Baby’s approval provides the necessary medicine optimized dose for the treatment of an otherwise neglected group of patients and offers a valuable addition to the antimalarial tool box.”
Dr. Marvel Brown, an associate professor at the Hertfordshire School of Health, Medicine and Life, says this should be seen as a major breakthrough in saving babies and young children.
“The mortality of malaria infections, especially in Africa of Subsahara, is extremely high – over 76% of deaths occur in children under five years of age.
“The increase in malaria death is further complicated in infants born with sickle cell disease, mainly due to a weak immune system.
“In terms of public health, Novartis, which makes this non -profit purpose, can help reduce inequality in access to healthcare.”